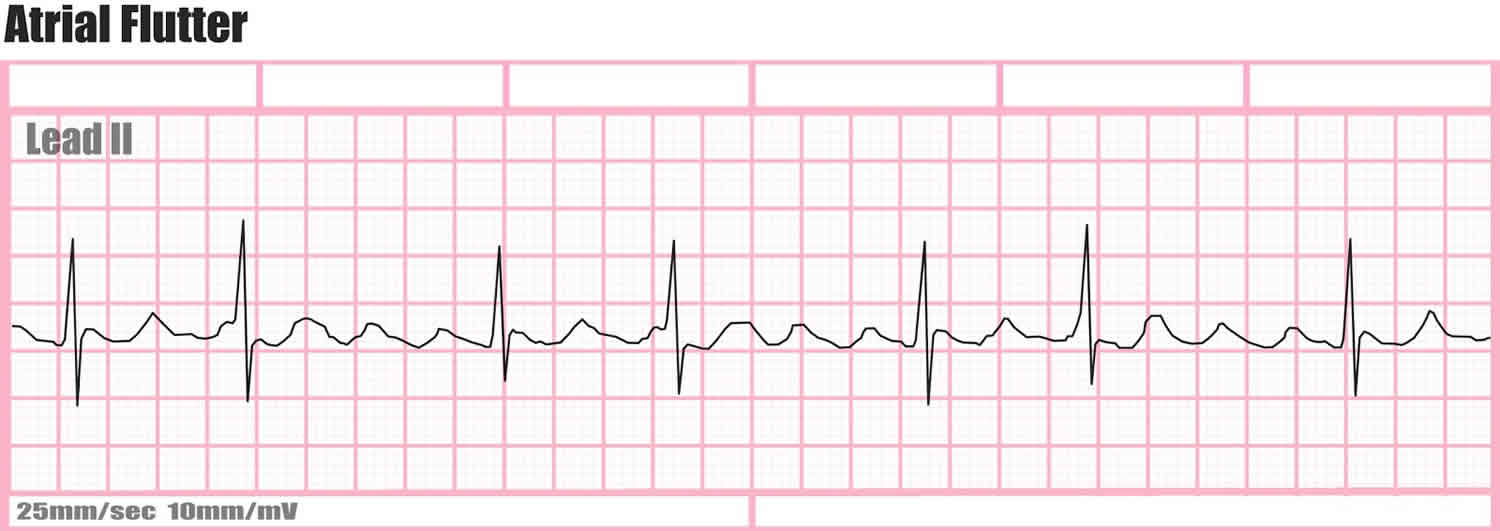
Furthermore, flecainide at low dose inhibits rapid component of the delayed rectifier K + current (I Kr) and at higher concentrations inhibits K ito channels. 
This review aims to highlight the main characteristics of flecainide, as well as its optimal clinical use, delineating drug indications and contraindications and appropriate monitoring, based on the most recent evidence.įlecainide works blocking the open-state fast inward Na + channel Nav 1.5 in a rate- and voltage-dependent manner, reducing the rise of phase 0 of the action potential especially in His-Purkinje tissue and ventricular muscle. 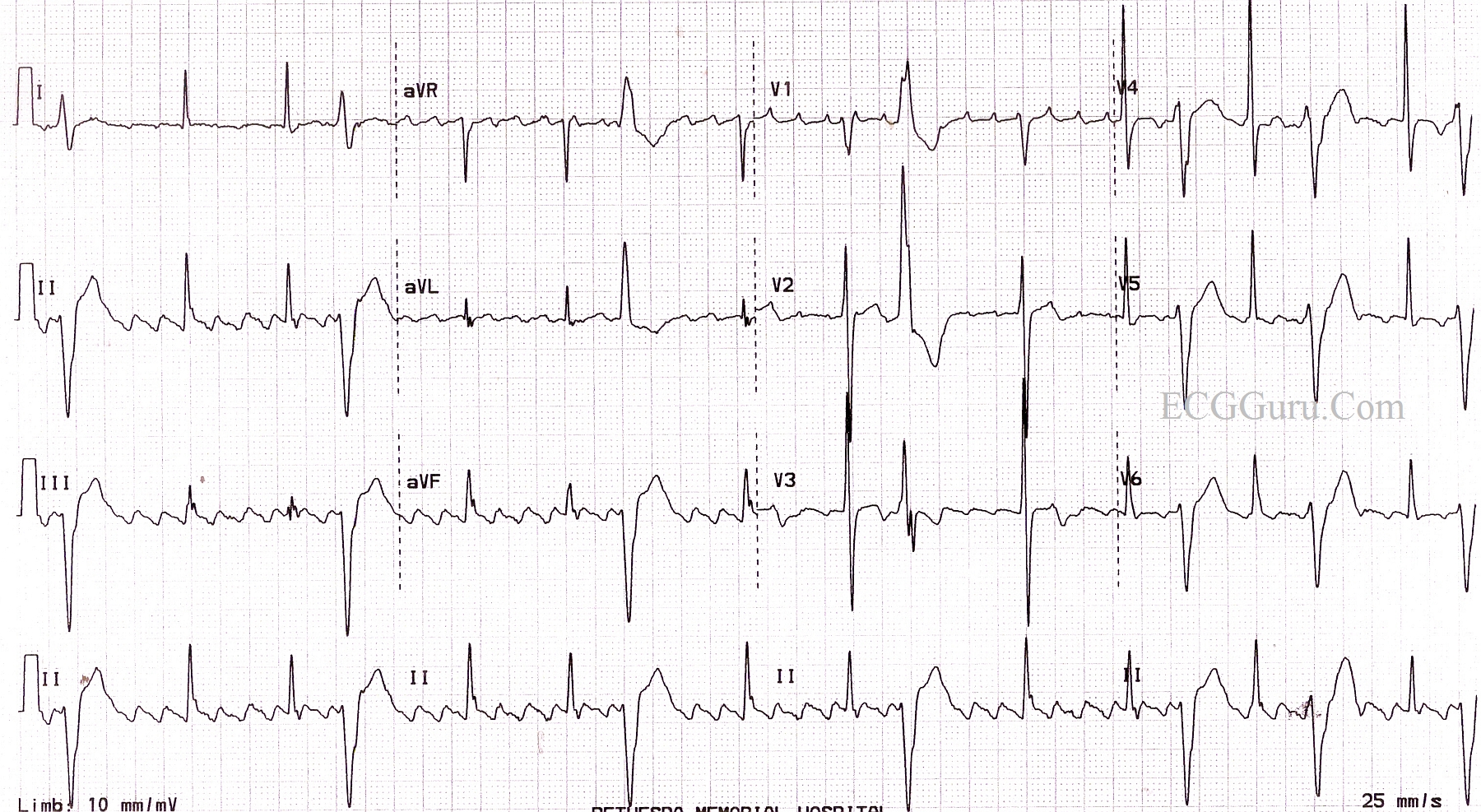
It is necessary to underline that many patients who died during the study had depressed left ventricular ejection fraction and intraventricular (IV) impulse disturbance, two conditions currently contraindicating the use of flecainide.ĭifferently from the CAST results, recent studies, enrolling different patient populations, have demonstrated a good safety profile of the drug combined with good clinical efficacy. The study was prematurely dismissed due to excess of mortality among patients treated with IC agents with a significant greater number of death and cardiac arrest due to arrhythmia than patients treated with placebo. 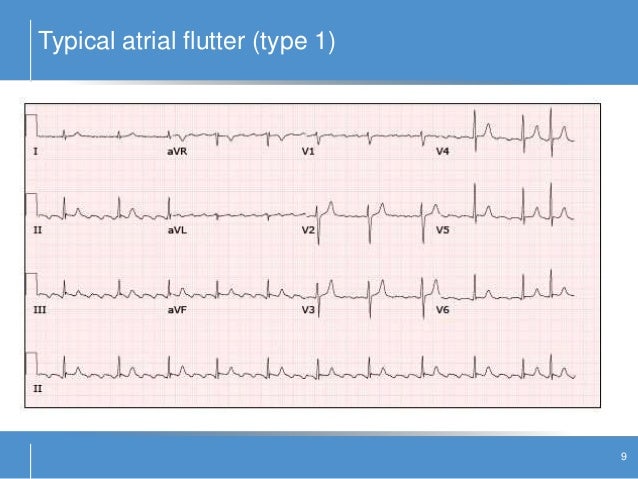
The CAST study published in 1991 has strongly conditioned and limited the actual use of flecainide in clinical practice. Nevertheless, antiarrhythmic drug treatment still plays a major role in patient management, alone or combined with non-pharmacological therapies.įlecainide is an IC antiarrhythmic drug approved in 1984 from the Food and Drug Administration for the suppression of sustained ventricular tachycardia (VT) and later for AF acute cardioversion and for sinus rhythm maintenance.Ĭurrently, flecainide is mostly administered for sinus rhythm maintenance and, having regard to its effectiveness and safety profile, it may be considered underused. In the recent years several non-pharmacological therapies, in particular transcatheter ablation, have been increasingly and successfully used to treat symptomatic drug refractory patients affected by supraventricular arrhythmias (SVT), especially atrial fibrillation (AF). How should we interpret the results of the CAST after the latest evidence? Is it possible to expand the indications of flecainide, and therefore, its use? This review aims to highlight the main characteristics of flecainide, as well as its optimal clinical use, delineating drug indications and contraindications and appropriate monitoring, based on the most recent evidence. Currently, flecainide is mostly used for sinus rhythm maintenance in atrial fibrillation (AF) patients without structural cardiomyopathy although recent studies enrolling different patient populations have demonstrated a good effectiveness and safety profile. Flecainide is an IC antiarrhythmic drug approved in 1984 from the Food and Drug Administration for the suppression of sustained ventricular tachycardia and later for acute cardioversion of atrial fibrillation and for sinus rhythm maintenance. Antiarrhythmic drug treatment still plays a major role in patient management, alone or combined with non-pharmacological therapies. Transcatheter ablation was increasingly and successfully used to treat symptomatic drug refractory patients affected by supraventricular arrhythmias.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
